Термодинамічні константи хімічних речовин
| Речовина | 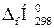 , , 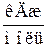
| 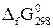 , , 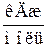
| 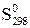 , , 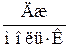
|
| Al2O3(корунд) | –1676,0 | –1582,0 | 50,92 |
| Al2S3(к) | –509,0 | –23,0 | 96,20 |
| Al(OH)3(к) | –1315,0 | –1157,0 | 71,10 |
| AlCl3(к) | – 704,6 | – 629,0 | 109,29 |
| AgCl (к) | – 126,78 | – 109,54 | 96,23 |
| BaCO3(к) | – 1217 | – 1137,2 | 112,13 |
| BaO(к) | – 553.9 | – 525,4 | 70,30 |
| CH4(г) | – 74,85 | – 50,85 | 186,27 |
| C2H2(г) | 226,75 | 209,21 | 200,82 |
| C2H4(г) | 52,30 | 68,14 | 219,45 |
| C2H6(г) | – 84,67 | – 32,93 | 229,49 |
| CH3COOH(р) | – 484,09 | – 389,36 | 159,83 |
| C2H5OH(г) | – 234,80 | – 167,96 | 281,38 |
| C2H5OH(р) | – 276,98 | – 174,15 | 160,67 |
| C6H6(р) | 49,03 | 124,38 | 173,26 |
| С6H12О6(к) | – 1273 | – 919,5 | 212,13 |
| CCl4(р) | – 132,84 | – 62,66 | 216,19 |
| CaBr2(к) | – 674,9 | – 656,1 | 130,00 |
| CaC2(к) | – 59,9 | – 64,9 | 69,96 |
| CaCl2(к) | – 796,3 | – 748,9 | 108,37 |
| CO(г) | –110,5 | –137,1 | 197,55 |
| CO2(г) | –393,5 | –394,4 | 213,66 |
| СаО(к) | –635,5 | –604,2 | 38,07 |
| CaCO3(к) | –1207 | –1129,6 | 91,71 |
| Ca(OH)2(к) | – 985,12 | – 897,52 | 83,39 |
| Ca3(PO4)2(к) | – 4123,6 | – 3887,6 | 235,98 |
| CuO(к) | –162,0 | –129,9 | 42,63 |
| Cu(NO3)2(к) | – 307,11 | – 114,22 | 191,30 |
| CuSO4(к) | – 770,9 | – 661,8 | 109,20 |
| CrO3 (к) | – 590,36 | – 513,44 | 73,22 |
| Cr2O3 (к) | – 1140,56 | – 1058,97 | 81,17 |
| FeCl3 (к) | – 399,70 | – 334,20 | 130,10 |
| FeO (к) | – 264,85 | – 244,30 | 60,75 |
| Fe2O3 (к) | – 822,16 | – 740,34 | 87,45 |
| Fe3O4 (к) | – 1117,13 | – 1014,17 | 146,19 |
| HBr (г) | – 36,38 | – 53,43 | 198,58 |
| HCl (г) | – 92,31 | – 95,30 | 186,79 |
| HF (г) | – 273,30 | – 275,41 | 173,67 |
| Речовина | 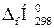 , , 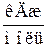
| 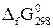 , , 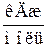
| 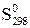 , , 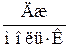
|
| HI (г) | 26,36 | 1,58 | 206,48 |
| H2O (г) | – 241,8 | – 228,61 | 188,72 |
| H2O (р) | – 286,0 | – 237,23 | 69,95 |
| H2O2 (р) | – 187,86 | – 120,52 | 109,60 |
| H2S(г) | –21,0 | –33,8 | 205,70 |
| KBr(к) | – 392,2 | – 379,2 | 95,94 |
| KCl (к) | – 436,68 | – 408,93 | 82,55 |
| KClO3(к) | – 391,20 | – 289,80 | 142,97 |
| KNO3(к) | – 492,46 | – 392,75 | 132,88 |
| MgCl2(к) | – 644,80 | – 595,30 | 89,54 |
| MgO (к) | – 601,8 | – 569,6 | 27,07 |
| NH3 (г) | – 45,94 | – 16,48 | 192,66 |
| NH4Cl (к) | – 314,22 | – 203,22 | 95,81 |
| NH4NO3(к) | – 365,43 | – 183,93 | 151,04 |
| (NH4)2SO4 | – 1180,0 | – 901,3 | 220,08 |
| NO(г) | 90,3 | 80,6 | 210,64 |
| NO2(г) | 33,0 | 51,5 | 240,06 |
| NiO (к) | – 239,7 | – 211,6 | 37,99 |
| NaBr (к) | – 361,41 | – 349,34 | 86,82 |
| NaCl (к) | – 411,12 | – 384,13 | 72,13 |
| Na2CO3(к) | – 1130,80 | – 1048,20 | 138,80 |
| NaNO3(к) | – 466,70 | – 365,97 | 116,50 |
| P2O5 (к) | – 1507,20 | – 1371,7 | 140,30 |
| Pb(NO3)2(к) | – 451,70 | – 258,90 | 217,90 |
| РbО (черв.) | –219,3 | –189,1 | 66,11 |
| РbO2 (к) | –276,6 | –218,3 | 71,92 |
| SO2 (г) | – 296,90 | – 300,21 | 248,07 |
| SO3 (г) | – 395,85 | – 371,17 | 256,69 |
| SnO2(к) | – 580,8 | – 519,3 | 52,30 |
| TiO2 (к) | – 943,9 | – 888,6 | 50,33 |
| ZnO (к) | – 348,11 | – 320,7 | 43,51 |
| ZnS(к) | – 205,6 | – 200,9 | 57,66 |
Примітка. Значення 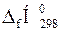 і
і 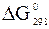 для простих речовин дорівнюють нулю;
для простих речовин дорівнюють нулю;
к – кристалічний, г – газоподібний, р – рідкий.
Додаток 2
Добуток розчинності при 25 0С(n – кількість йонів)
| Сполука | ДР, (г-йон/л)n | Сполука | ДР, (г-йон/л)n | Сполука | ДР, (г-йон/л)n |
| AgBr AgCN AgCl AgI AgBrO3 Ag2CrO4 Ag2SO4 Al(OH)3 Ag2S BaSO3 BaSO4 | 5,0·10-13 1,6·10-14 1,7·10-10 8,1·10-17 6,0·10-5 4,4·10-12 1,2·10-5 4,0·10-33 2,5·10-25 9,5·10-10 1,5·10-9 | Ca(OH)2 CaSO4 Cd(OH)2 Co(OH)2 CuCl CuІ Fe(OH)2 Hg2Br2 Hg2Cl2 Hg2І2 HgS | 6,0·10-6 2,4·10-5 1,7·10-14 2,5·10-16 3,2·10-7 1,1·10-12 1,4·10-15 4,0·10-23 1,0·10-18 4,0·10-29 1,6·10-52 | Hg2SO4 Ni(OH)2 PbBr2 PbСl2 PbІ2 Pb(OH)2 PbSO4 TIBr TICl Zn(OH)2 ZnS | 6,2·10-7 1,3·10-16 4,5·10-6 1,5·10-5 8·10-9 5·10-16 1,6·10-8 3,6·10-6 1,8·10-4 4,3·10-17 1,3·10-23 |
Додаток 3